The surge in Covid cases and deaths in India in April led to record highs for the global number of daily cases, and only the under-counting of deaths is likely to have meant that there were not also record global highs for daily deaths. This pandemic is far from over, even if it is starting to seem like that to the high-income countries that make up the G7.
The UK G7 summit must take decisive action to bring the pandemic to an end. This is both the right thing to do to demonstrate solidarity and common need, and in the self-interest of the G7. Continuing high levels of infection around the world create the risk of new variants emerging, putting at risk the gains made elsewhere. Delay in getting the pandemic under control globally is also economically costly – to all countries. The costs of vaccinating the world pale in comparison. Low and middle-income countries do not have the same fiscal capacity to address the pandemic, and as a result are projected by the IMF to have the slowest economic recovery across all income groups.
The G7 cannot on its own solve a global pandemic, but it can be a major part of the solution by taking the following actions:
- Meet the full funding needs of the ACT-Accelerator for therapeutics, diagnostics and PPE and dramatically expand the ambition and level of financing of COVAX to target the vaccination of all adults in the 92 mostly low- and middle-income countries which are eligible for its support.
- Provide support for countries to roll out their vaccination campaigns, and to purchase vaccines from other pooled platforms such as the African Union’s Africa Medical Supplies Platform. The total additional financial support needed across these two areas is likely to be around $34 billion–$40 billion depending on the price at which vaccines can be purchased. Around $24–$30 billion of this will need to be new grant funding, with the remaining $10 billion met by the existing World Bank commitment to provide $12 billion of support for vaccine purchase and roll-out (which covers a broader set of countries), and countries’ own funding. G7 countries should commit to funding around two-thirds of this, and coordinate with other OECD DAC donors to provide the rest.
- Maximise global vaccine manufacturing capacity by agreeing to a temporary and limited waiver on compulsory licensing to allow the export of vaccines from any manufacturing facility to any country in need, and the removal of restrictions on parallel imports, backed by compensatory payments to holders of intellectual property rights. And take coordinated action to remove bottlenecks in pharmaceutical supply chains.
Significantly increased financing commitments
Peter A. Singer, Special Adviser to the director-general of the World Health Organization, stated in January that ‘vaccine equity will be the defining challenge of 2021’ as ‘none of us will be safe until we are all safe’. As the locus of the pandemic has shifted to the global south, the failure to adequately prioritise vaccine equity is becoming increasingly apparent (Figure 1).
Figure 1: New deaths 7-day rolling average, by region
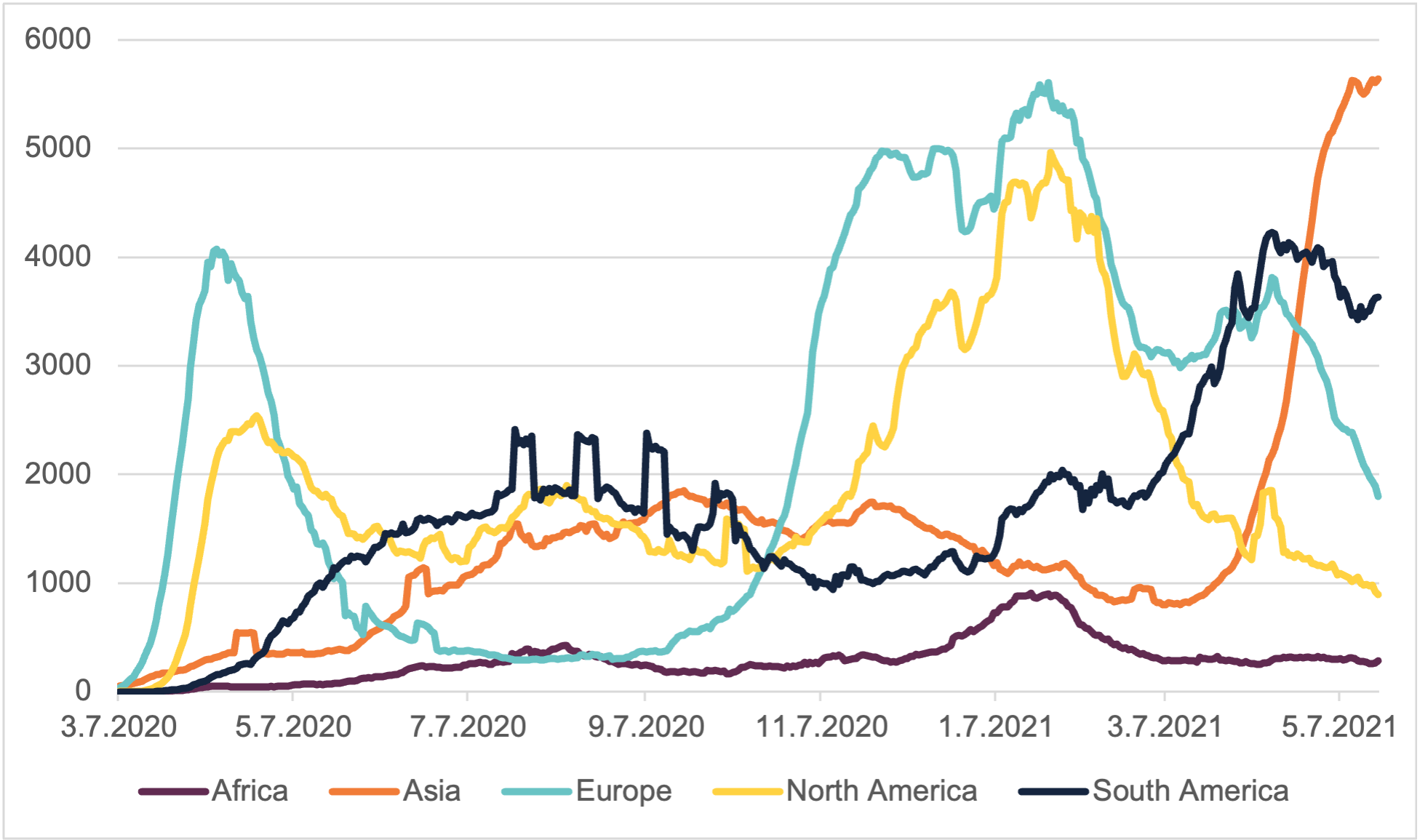
As of 9 May 2021, 44% of vaccinations had been given in high-income countries representing only 16% of the world’s population. The nearly half of the world’s population who reside in low and lower middle-income countries has received 17% of vaccinations. While some high-income countries have made rapid progress in vaccinating their populations, many low-income countries have barely started (Figure 2).
Figure 2: Vaccine coverage by income level
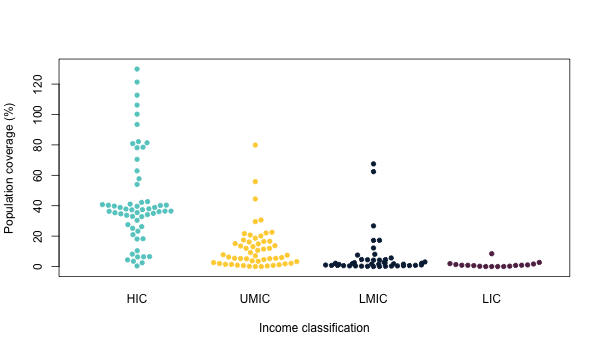
Note: full coverage is achieved at 200%, when the whole population have had two shots.
The main global mechanism for supporting the roll-out of Covid-19 vaccines is the COVAX Advance Market Commitment (AMC) facility, which will procure vaccines for 92 mostly low and lower-middle income countries. This has currently received financial commitments of $9.1 billion, which would enable it to procure around 1.8 billion doses, enough to immunise around 30% of eligible countries’ populations.
However, even a target of financing 30% of population coverage for immunisation in 2021 seems astonishingly unambitious. Most high-income countries are targeting the vaccination of all adults aged over 18 – on average nearly 80% of their populations – well before the end of 2021, and are already licensing vaccines for use for teenagers. The target of 30% is also far below the targets countries themselves are setting. COVAX has reported that the average target population coverage for 45 countries who have reported this is nearly 50% and nearly all AMC-eligible countries have requested more vaccines from COVAX than it can supply in 2021. Africa CDC has recommended that African countries aim to vaccinate 60% of their populations.
Yet the pace of international support means that, in many low and middle-income countries, it could be well into 2023 before such coverage is achieved. As a result of slow progress in vaccine distribution via COVAX, low- and middle-income countries are also seeking to procure their own vaccines, despite their fiscal challenges. The African Union and the African Export-Import Bank have arranged a $2 billion advance procurement commitment for 270 million vaccine doses that will cover around 15% of Africa’s population. Together, the Africa CDC and the existing COVAX orders could thus help Africa’s low and lower middle-income countries reach around 45% of their populations.
There is a three-fold case for committing further international funding. First, it is in the self-interest of high-income countries to support a more rapid global immunisation campaign. The spread of variant B.1.617 is putting the UK timetable for the easing of lockdown measures at risk. If the pandemic is not contained in the global south, this puts at risk the gains made in high-income countries. Second, delay is economically costly. The IMF estimates that the benefits of a faster end to the pandemic could amount to a $9 trillion gain for the global economy. The costs of ensuring greater vaccination coverage, as we set out below, are orders of magnitude lower, measured in the billions, not the trillions.
Third, it is the right thing to do to support poor countries, which are projected to have the slowest economic recovery across all income groups, and do not have the same fiscal firepower as richer countries. As the managing director of the IMF, Kristalina Georgieva, points out, vaccine policy is economic policy. Delayed distribution of vaccines could set back even further the recovery of low- and middle-income countries – precisely those with the least resources to cope with the twin disease and economic shocks of Covid-19. The World Bank forecasts an unprecedented reversal in the trend of declining poverty, with more than 100 million people being pushed into extreme poverty by the crisis.
Increased funding should not only scale up the ambition and reach of COVAX, but also give a strong financial signal that there is guaranteed demand, so that investment in vaccine manufacturing capacity takes places as quickly as possible. It should also fully fund the ACT-Accelerator to improve access to diagnostic tests and therapeutics, such as oxygen and PPE. The diagnostics pillar of the ACT-Accelerator currently has the largest funding gap ($8.7 billion), yet testing is essential to track the spread of the pandemic, and for countries to implement effective containment strategies.
What would such financial commitments look like?
First, all the pillars of the ACT-Accelerator should be fully funded. The therapeutics, diagnostics and health systems connector pillars have a total shortfall of $19.9 billion. Conservatively assuming that all of the ACT-A pending allocation of $3.3 billion is allocated to these pillars, and none to COVAX, leaves a funding gap of $15.9 billion across these three pillars.
Second, COVAX should be provided with sufficient assured funding to enable it to reach the entire adult population in eligible countries. This is around 2.4 billion people across 92 countries – or on average 62% of their adult population. This would cost an around an additional $7.1 billion on top of the existing $9.1 billion that has already been pledged, and which COVAX estimates would allow it to reach 30% of the population. This is based on the assumption that COVAX can continue to procure vaccines for around $3 a dose, and a 10% vaccine wastage rate. If this increased to $5.25 a dose this would cost around $12.5 billion, or $5.4 billion more. These figures exclude the vaccines secured by the African Union for around 15% of African countries’ populations.
The third financial commitment is thus to provide African countries with funding to purchase from this pooled mechanism, and to provide support for vaccine roll-out costs. On top of the approximately $2 billion for African countries to purchase vaccines, the cost of rolling out vaccination to all adults in the AMC 92 countries would be around $8.1 billion, based on the country-level data in Diab et al. (2021). However, such estimates do not include the cost of hiring additional health workers, which is very likely to be needed in such a large scale-up. Assuming that countries will bear some of the roll-out costs themselves, this funding would ideally be secured from the $12 billion the World Bank has made available to support countries to purchase and roll out vaccines. Other technical assistance and monitoring could cost around $1.2 billion based on estimates reported by the COVAX working group on delivery costs. This totals an additional $34 billion of support, of which $24 billion will need to be new commitments, assuming that existing World Bank commitments and countries’ own funding can cover $10 billion.
How could this be funded?
Norway has proposed that financial contributions to ACT-A be based on the IMF quota formula, adjusted for GDP per capita. This would require larger, richer economies to make larger commitments, but proposes that all major economies make contributions. Such a contribution system is utilised here to suggest what major economies could contribute to make the additional $24 billion support available. As so far no middle-income economies have made significant contributions to ACT-A, we assume that the financing needs outlined above will only be met by DAC members, which will scale up their contributions proportionally. MICs that have not made contributions to ACT-A but which are included in the Norwegian proposal include China, India, Brazil, the Russian Federation, Mexico, Turkey, Indonesia and Thailand. As well as these MICs, two high-income countries that have not yet made contributions and which have proposed contributions of <1%, equating to around $350 million at the funding levels we propose here, are Poland and United Arab Emirates. Countries such as Norway and Saudi Arabia, which have already made large donations, would only need to increase funding by the relatively small amount of $50–90 million. By contrast, the US would need to increase funding by $8.7 billion, Japan by $3 billion, France by $1.7 billion and the UK, South Korea, Italy, Australia and Switzerland by around $1 billion. As Germany has already made a large commitment of $2.5 billion it would only need to increase this by around $570 million.
This additional financing is not about providing aid – even if much of this support could be formally counted as ODA under current rules – it is about providing the extraordinary investment needed to help the world recover from Covid-19. Providing this funding, with the UK playing a lead role in coordinating this as chair of the G7, is the pragmatic thing to do.
What role could providing vaccinations in kind play?
To vaccinate the 2.4 billion or so adults in the AMC 92 countries, COVAX will need around 5.4 billion doses. It has already procured around 2.1 billion doses for these countries, leaving a gap of 3.3 billion. According to Duke Global Health Innovation Center’s procurement tracker, some high-income countries have ordered many more doses than they need. Between them, the EU, US, UK and Canada have ordered over 3 billion doses for a total population of 880 million. Overall, Duke has tracked orders of 11.6 billion doses of vaccine, although not all of these will be delivered in 2021. This suggests that, with prioritisation, vaccines could be made available for most of the 5.4 billion adults in the world, if not by the end of 2021, then early in 2022.
How can we scale up production?
A key challenge is production capacity. This has led to some countries, and the COVAX facility itself, not receiving all the vaccines they need. UNICEF has reported that COVAX is nearly 200 million doses short of what it expected to receive in May and June as a result of India’s Serum Institute stopping exports to meet domestic needs. The commitment of the US and EU to donate 180 million doses between them is welcome, but inadequate because not all of these donations will be provided to COVAX, and they are unlikely to arrive by June.
Duke’s latest manufacturing projections suggest over 11 billion vaccines will be produced in 2021. However, manufacturers’ projections may well be optimistic. Providing COVAX with the funding for vaccines for all adults in AMC 92 countries would demonstrate that there is clear demand for greater manufacturing capacity, not just in high-income countries but across the globe. It will also require action to boost the use of existing production capacity anywhere in the world. With most leading pharmaceutical production facilities working at full capacity in rich economies, it will be necessary to look for third-party or government-owned capabilities elsewhere, typically in middle-income countries. The G7 must engage with, and support, countries where these capabilities are present to coordinate the effort and boost vaccine availability.
A temporary and limited waiver on compulsory licensing (that under the WTO TRIPS agreement allows governments to issue licenses to supply exclusively the domestic market) needs to be considered to allow the export of patented and licensed vaccines from third-party facilities anywhere in the world to poorer countries where these production capabilities do not exist. In this way, unused capacity in many of these facilities can be used to speed up the worldwide roll-out of existing vaccines. Moreover, any restrictions that members may have in regard to parallel imports must be temporarily eliminated to avoid any possibility of price discrimination.
It is also critical that the developers of the vaccines under compulsory license remain involved, to guarantee the quality and effectiveness of the products, and that they are remunerated for the property rights under compulsory licensing provisions. The G7 and other partners could provide the funding for these payments.
As well as seeing that worldwide manufacturing capability is maximised, the G7 should ensure that any bottlenecks in the supply chain of the pharmaceutical sector are addressed. Regulatory and institutional barriers restricting trade in any relevant input should be reduced or eliminated. The G7 should also aim to coordinate, and eventually support, the use of existing worldwide production capacity for these inputs.


